News

Ion Exchange Chromatography with Agarose Beads in Antibody Purification
ABT agarose beads provide reliable performance in ion exchange chromatography, ensuring high protein binding, precise separation of charge variants, and reproducible results from lab-scale analysis to industrial antibody purification.
Read more
ABT at Analytica 2026: Advancing Bioprocessing Innovation in Munich
From March 24–27, ABT will be showcasing its expertise in agarose-based resins and custom chromatography solutions at Analytica 2026 in Munich, Germany. Visit us to explore how our advanced purification technologies can support scalable and high-quality bioprocess workflows. Our team will be on-site to discuss tailored solutions designed for pharmaceutical and biotech leaders seeking robust, reproducible, and high-performance results.
Read more
ABT to Attend Bioprocessing Summit Europe 2026: High-Performance Agarose Resins for Downstream Processing
ABT will attend Bioprocessing Summit Europe 2026 in Barcelona. Discover how our agarose chromatography resins support scalable, robust downstream processing from early development to GMP manufacturing.
Read moreNext Stop: BPI Boston 2025!
ABT returns to Bioprocess International Boston, where the future of bioprocessing from cell line and cell culture to downstream and next generation manufacturing, is showcased.
Read more"This study highlights the importance of protein purification techniques to identify protein-protein interactions that are critical for cell biology"
The E3 ubiquitin ligase RNF2 protects polymerase ι from destabilization and represents a potential therapeutic target for diseases associated with DNA instability.
Polymerase ι (Polι) is a Y-family DNA polymerase involved in translesion synthesis (TLS), a critical mechanism for DNA damage tolerance. Its regulation is essential because its instability can compromise genome stability and lead to diseases such as cancer.
This study explores the role of RNF2, an E3 ubiquitin ligase, in protecting Polι from proteasomal degradation. For this purpose, co-immunoprecipitation (Co-IP) experiments were conducted using a glutathione agarose resin column from Agarose Beads Technologies (ABT) to purify the protein and evaluate the interaction between Polι and RNF2.
The stability of Polι was also analyzed in cells with and without RNF2, revealing that the loss of RNF2 resulted in a significant reduction of Polι due to proteasomal degradation.
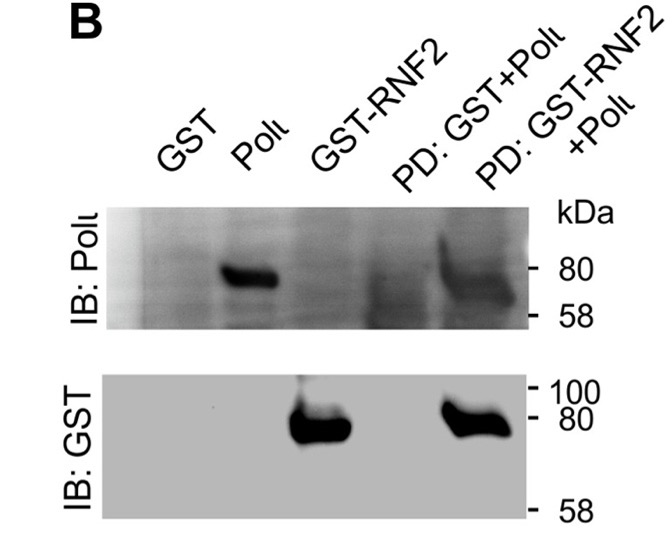
Figure 1. Polɩ interacts with E3 ubiquitin ligase, RNF2. (B) Pull-down assay showing the interaction between GST-tagged RNF2 and His-tagged Polɩ. His-Polɩ was incubated with GST- or GST-RNF2. GST was captured on glutathione agarose beads, washed and separated by SDS-PAGE parallel to 5% of protein input, followed by immunoblotting with polyclonal antibodies raised to the C-terminus of Polɩ [37] and anti-GST antibodies (Covance and Santa Cruz, respectively)
The results indicate that RNF2 directly interacts with Polι and prevents its degradation, ensuring its function in translesion synthesis and reducing cellular susceptibility to DNA damage.
This study concludes that the regulation of Polι by RNF2 is critical for genome stability and suggests that it may represent a potential therapeutic target for diseases associated with DNA instability.
Furthermore, it highlights the importance of protein purification techniques using ABT agarose beads to identify protein-protein interactions that are critical for cell biology.
- Fedorowicz M. et al., IE3 ubiquitin ligase RNF2 protects polymerase ι from destabilization, Molecular Cell Research (2024).





